Coronavirus / COVID
Latest News
Latest Videos

CME Content
More News

A new study found people who staunchly oppose COVID-19 vaccines were more likely to have experienced adverse childhoods, making them distrustful from a young age.
Notoriously slow to implement new practices, most hospitals rapidly updated their standard treatment procedures during the COVID-19 pandemic.

A pair of new studies in the Journal of the American Medical Association add to growing evidence that COVID-19 vaccines are safe during pregnancy, showing no increased risk for adverse outcomes among women who were vaccinated during pregnancy and their babies.

Children under 5 became infected with the Omicron variant 6-8 times more frequently than young children who contracted Delta. However, Delta COVID-19 infections were more severe than Omicron.
Here is a rundown of the most popular stories we covered this past week.

Despite lower comorbidity risk scores than Black or White COVID-19 patients, Mississippi’s Indigenous populations had significantly higher in-hospital mortality rates.

One day after the FDA approved second booster shots for certain vulnerable populations, the National Institute of Health (NIH) announced they have begun enrolling adult US participants in a phase 2 clinical trial to evaluate a second COVID-19 booster dose.
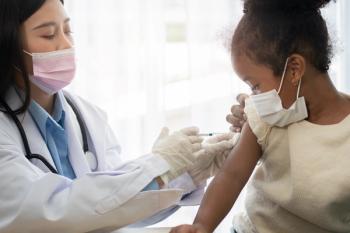
2 doses of the Pfizer-BioNTech vaccine reduced Omicron hospitalizations by 68% in children 5-11 and by 40% in adolescents 12-18 years old.

The Pfizer-BioNTech and Moderna mRNA vaccines were found to produce different antibody and killer T-cell responses, suggesting a “mix and match” booster approach may provide the best protection against COVID-19.
“Together these 2 diseases create a perfect storm”: Black people with cancer were more likely to have severe or fatal COVID-19 disease.

New policy appears inconsistent and, on the surface, unfair, expert says.

The new action by the federal agency now means the additional booster shot can be administered to people 50 years and older.
The coronavirus pandemic caused the first global recorded decline in human life expectancy.

The Assistant Secretary for Preparedness and Response (ASPR) announced a pause in sotrovimab distribution in regions where BA.2 is the dominant COVID-19 variant, citing evidence that the monoclonal antibody therapy would not effectively neutralize the BA.2 Omicron variant.

The federal agency is expected to provide the EUAs prior to their next VRBPAC meeting on April 6.
Check out the important stories we covered this week.

People with immune dysfunction are at increased risk for COVID-19 breakthrough infection after vaccination, and should take additional precautions.

The company is developing a single combination vaccine to cover SARS-CoV-2, influenza, and respiratory syncytial virus (RSV) as well as another candidate for endemic human coronaviruses.

This designation of the company’s respiratory syncytial virus (RSV) vaccine candidate, PF-06928316, will help to expedite its development and review.

The findings are significant given that a second booster dose may soon be recommended.

Research set to be presented next month found that people with resolved COVID-19, and in some cases active COVID-19, could safely donate organs.

Along with reporting positive data in the youngest pediatric population, the company is working with the Food and Drug Administration (FDA) for emergency use authorization of its vaccine in children 6 years to under 11 years as well as the 12-17 age groups.

COVID-19 vaccines were less protective against symptomatic infection from the Omicron COVID-19 variant than the Delta variant.

The Omicron variant of SARS-CoV-2 had a secondary attack rate of 25.1% in Norwegian households compared with 19.4% for the Delta variant.

A matched cohort study found COVID-19 infection increased the risk of new type 2 diabetes diagnosis. Compared to patients with acute upper respiratory tract infections, COVID-19 patients were 28% more likely to develop diabetes.