C. Difficile
Latest News

Latest Videos

CME Content
More News
DDW 2024 highlighted advances in managing recurrent C difficile infections, emphasis on the effectiveness of live biotherapeutic products and the impact of antibiotic stewardship, and more ongoing efforts in combating C difficile.

The FDA recalls ice cream, orzo, and mixed nuts. Also this week, interviews with Jared Baeten, MD, PhD, on Lenacapavir's 100% efficacy against HIV, and Jason Roberts, PhD, on the promise of continuous β-Lactam against sepsis, and more from Contagion.
A Florida hospital takes a novel approach for the healthcare-associated infection and reduced inappropriate testing by setting up guidelines.

A case study of an 80-year-old patient, illustrating successful management of dual H pylori and C diff infections with quadruple therapy, with key insights from Yazan Abboud, MD and Sima Vossough-Teehan, MD.
Researchers assess the efficacy of fecal microbiota transplantation (FMT) for first or second episodes of Cdifficile infection (CDI), demonstrating high initial cure rates and sustained in managing the condition.
In a small study, recurrent Clostridioides difficile infection (CDI) therapy, Rebyota, was durable in a majority of patients who were exposed to non-CDI antibiotics.

FDA approves pneumococcal vaccine V116, Salmonella outbreak linked to a household pet, Japan is currently facing an unprecedented surge in cases of streptococcal toxic shock syndrome, and more this week from Contagion.

In a 17-year study across 2 Québec hospitals, researchers explored the relationship between antibiotic use and healthcare-associated Clostridioides difficile infections.

Presented at DDW 2024 by Ken Blount, the effectiveness of live biotherapeutic products in treating recurrent C difficile infections (rCDI), offering insights into microbiota alteration and prospects for management.
The link between vancomycin susceptibility and decreasing sustained clinical response in C difficile infections.
Minnie Hatch was in her late teens when she was diagnosed with the infection. Since then, she has had recurrent episodes and dealt with care challenges along the way.
Vedanta Biosciences initiates the Phase 3 RESTORATiVE303 clinical trial, marking a pivotal moment in the battle against this recurrent infection.

This week, a strategic approach along with existing treatments aimed at reducing incidence rates and curing people of HCV, advocating Lyme disease research in honor of May being its awareness month, Digestive Disease Week coverage, and more this week from Contagion.

Antimicrobial stewardship to reduce multidrug resistance and opportunistic Clostridioides difficile infection (CDI) could also guide shorter treatment.

This week, new WHO data illustrates challenges during the acute phase of the COVID-19 pandemic, MAD-ID and ACVR conference coverage, and more from Contagion.
In patients with Clostridium difficile infection (CDI), ibezapolstat was shown to have similar results to a standard of care therapy.

Insights from Dr. Md Zahidul Alam on treatments and prevention strategies for C difficile infection (CDI), supported by the CDC's recent study on CDI management in healthcare settings.
Check out the latest studies, management strategies, patient advocacy, and other important news involving this condition.
A study presented at ESCMID aims to understand ribotype 955 as a new challenge in C difficile management.
A study presented at the Society for Healthcare Epidemiology of America (SHEA) conference assesses the link between antibiotic use and C difficile infection in Tennessee.
A study, presented at the Society for Healthcare Epidemiology of America (SHEA) conference, assesses laxative administration before CDI testing in Veteran Affairs (VA) hospitals and explores the demographic and contextual factors with non-adherence to guidelines.

Incidence of hospital-onset Clostridioides difficile infection differed between hospitals that implemented the CDC prevention strategies and those that did not, but not necessarily because of the strategies.

This annual event hosted by The Peggy Lillis Foundation (PLF) released their full agenda for advocacy and education that will be happen next week in Washington DC.
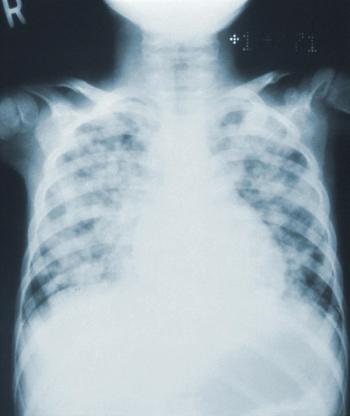
Community acquired pneumonia is often inappropriately diagnosed in hospital admissions, particularly among older adults with dementia, and can lead to in-hospital antibiotic-associated adverse events, such as Clostridioides difficile.
A study on the impact of guidelines on reducing hospital-onset CDI incidence.