Since the vancomycin guideline update, institutional leaders have been faced with the challenge of implementing and monitoring AUC-guided dosing.

Since the vancomycin guideline update, institutional leaders have been faced with the challenge of implementing and monitoring AUC-guided dosing.

Despite effective vaccines and other prevention and treatment modalities, hepatitis B remains a global health challenge. “It’s not for the lack of available tools,” says professor and hepatologist H. Nina Kim, MD, MSc.
Carbapenem-resistant Enterobacterales present considerable treatment challenges. Recent cumulative evidence supports preference for therapies.

“Patients deserve more than just viral suppression,” said Harmony Garges, MD, chief medical officer of ViiV Healthcare.

The federal agency set a Prescription Drug User Fee Act (PDUFA) action date of August 2023.
A researcher who presented at CROI discusses this prolonged and debilitating condition.
A clinician presenting at CROI discusses the phenomenon and offers insights and considerations for treating patients who are dealing with this.
The unidentified German man cleared the virus after hematopoietic stem cell transplantation (HSCT), and seems to have eliminated the HIV reservoir, which would be further along than the first two HIV-free patients who underwent the same procedure.

Results reported at CROI showed that 2 cohorts of the ongoing, phase 3 DELIVER study that this form of PrEP modality posed no differences in pregnancy and infant outcomes.

“We should test everybody,” said Charles Béguelin, noting that the vast majority of hepatitis D infections go undetected.

“One size never fits all,” says hepatologist Anna Suk-Fong Lok, MD, emphasizing the need to tailor hepatitis B treatment to fit the patient.

Inequities can include access to care issues that run from prevention to acute treatment. Addressing such issues can lead to better outcomes.

A clinician offers insights on the initial conversations around learning to treat a chronic disease and the importance behind getting patients started down the right treatment path in the early days following a diagnosis.

With evolving weather patterns, the natural habitat of ticks is expanding, and more people are becoming susceptible to them.

Although fecal microbiota, live-jslm (Rebyota; RBX2660) is the first fecal microbiota product approved by the FDA, there are other microbiota-based agents in the pipeline.

The agency accepted 2 Supplemental New Drug Applications (sNDA) related to 2 different transplant populations.

Both undertreatment and overtreatment were common in this cohort of penicillin-allergic pregnant women with Group B Streptococcus (GBS).

A recent study suggests that metabolic factors such as hypertension and diabetes were associated with an increased risk of liver fibrosis among patients with metabolic associated fatty liver disease who had hepatitis B virus infection.

Extended infusion of the antibiotics resulted in fewer patients who died, a shorter length of hospitalization, and lower incidence of C difficile.

This study characterized the incidence of serotonin syndrome and identified contributing risk factors with combined medications for opioid use disorder (MOUD) and linezolid use.

Administered during the Omicron-dominant phase, the vaccine's efficacy was 73.2% in individuals 6 months to 4 years of age.

Why have “long COVID” cases become less frequent and less severe over time?

Although HIV remains a pervasive epidemic, there have been tremendous advancements and progress in HIV prevention, and the work continues.

Ferring’s RBX2660 can be utilized by clinicians for patients with recurrent C diff.

Here is an overview of highly anticipated HIV trial data ViiV Healthcare will share next week at the CROI 2023 conference.

Some chronic hepatitis B patients experienced a dramatic decrease in hepatitis B surface antigen (HBsAg) after receiving a COVID-19 vaccine. Could there be a correlation?
A hospital reviewed both treatments in a real-world clinical setting to determine the effectiveness in preventing early and late recurrences.
Norovirus infections are spiking this season. These are the signs and symptoms associated with America’s leading cause of foodborne illness.

Six respiratory infections are among the most common diagnoses for which antibiotics often are inappropriately prescribed, and new research sheds light on the effects, both in increased risk of adverse events and excess health care costs.
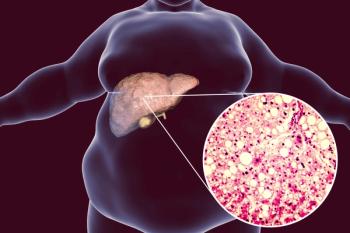
This finding, in addition to published clinical data, suggests VEGF-B signaling may play a significant role in human non-alcoholic fatty liver disease pathophysiology.