Initially authorized based on promising trial data, recent analyses raise questions about the effectiveness of nirmatrelvir/ritonavir and molnupiravir against new viral variants, particularly regarding mortality and hospitalization rates.

Initially authorized based on promising trial data, recent analyses raise questions about the effectiveness of nirmatrelvir/ritonavir and molnupiravir against new viral variants, particularly regarding mortality and hospitalization rates.
A large study shows a moderate benefit to taking the antimalarial therapy.

By analyzing 38 strains from patients, researchers identified clusters linked to frequent room changes, highlighting the need for improved infection control measures.

New predictive statistical modeling shows that antimicrobial resistance (AMR) deaths will rise steadily in the coming decades.
The model predicts and stratifies hepatocellular carcinoma risk by analyzing serum HBV DNA levels and other factors to enhance early detection and management.
Adults with type 2 diabetes who use metformin have a 13% to 21% reduced risk of developing Long COVID or dying from COVID-19 compared to those taking other diabetes medications.
The CSL and Arcturus Therapeutics’ vaccine, ARCT-154 (Kostaive), is designed to offer protection against the JN1 strain.

Uzma Syed, MD, DO discussed how various factors increase women’s susceptibility to AMR, the importance of policies, and the need for culturally specific education and advocacy to improve health outcomes.

The International Society for Infectious Diseases (ISID) ProMED surveillance system is a way for clinicians and interested ID stakeholders to be aware of local outbreaks and follow-up in response to get more information on them.
With no vaccines or specific treatments available, the mosquito-borne pathogen, which causes fever, severe headache, and joint pain, raises alarms due to its spread across the Americas.

A recent study showed that an increase in anti-hepatitis E virus seroprevalence in patients with chronic inflammatory demyelinating polyneuropathy was not a consequence of intravenous immunoglobulin therapy.

The organization's designation for the Bavarian Nordic’s MVA-BN (Jynneos) mpox vaccine is aimed at helping with the public health response, especially in Africa, where incidences rates continue to increase.

Overall, findings demonstrated a 78% treatment success rate among patients who prematurely discontinued direct-acting antivirals (DAA), and even seeing a 93% success rate among those who discontinued after week 4.

This week, World AMR Congress interviews, Long COVID’s toll on the brain, e coli outbreak linked to lettuce, and more.

At the World AMR Congress, Yann Ferris discussed how leveraging partnerships enhances the fight against antimicrobial resistance.

Ami Patel, PhD, provides insights on both technologies and how her research center, the Wistar Institute, is involved in DNA vaccine and monoclonal antibody research.
Protein organization could pave the way for effective vaccine development

Females are more frequently affected by Long COVID than males, highlighting distinct immunological processes and hormonal variations.

Gilead’s HIV-1 capsid inhibitor, which is a twice-yearly injection, was more effective than a daily Truvada pill in a phase 3 trial.
C diff remains a major healthcare burden globally, with over 225,000 hospitalizations in the US alone. Over half of these cases had recent antibiotic exposure, underlining the significant relationship between antibiotic use and the risk of the healthcare-associated infection.

The benefits, challenges, and prospects of wastewater data in enhancing public health strategies against AMR.

Crestone’s novel therapy, CRS3123, demonstrated 97% clinical cure rate in small phase 2 study.

The study on the E coli outbreak in the UK highlights the growing impact of climate change on public health and food security.

Kevin Nguyen, PharmD, offers some insights on some of the challenges in the outpatient setting with regards to stewardship, and novel strategies being utilized.
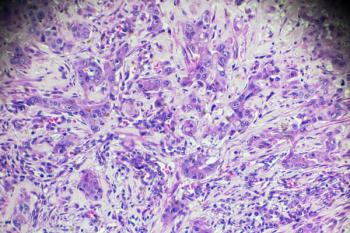
The gene USP26 is identified as a factor in hepatocellular carcinoma development and a potential therapeutic target.

Recent data and conversations with experts highlighted the increasing concern of Long COVID’s impact on the brain in patients who have been previously infected with the virus.

Internal tremors are a newly recognized and troubling symptom of Long COVID, experienced by over one-third of participants in a Yale-based study

Syra Madad, DHSC, MSc, MCP, CHEP, outlines early surveillance systems and diagnostic innovations for enhancing pandemic management and response.

Our panel discusses this patient group and the challenges associated with their care.

FDA announced the outbreak already impacted 65 people across 9 states, with 24 hospitalizations.