Vaccines
Latest News
Latest Videos

CME Content
More News

Following VRBPAC recommendations earlier this week, both the Pfizer-BioNTech and Moderna COVID-19 vaccines were FDA-authorized for children as young as 6 months.

At separate press conferences, the CDC and WHO indicate that containing monkeypox will depend on testing, tracing, and currently available vaccines.

Today, the FDA's Vaccines and Related Biological Products Advisory Committee (VRBPAC) unanimously voted to recommend both the Moderna and Pfizer-BioNTech COVID-19 vaccines for children as young as 6 months old.

The committee voted twice splitting the questions into younger children (6-11 years) and adolescent (12-17 years) cohorts due to different vaccine dosing amounts.

Sanofi and GSK’s booster vaccine candidate is modeled on the Beta COVID-19 variant, and increases Omicron-neutralizing antibody titers 40-fold.

Data results from its phase 3 trial showed it was statistically significant and the company plans to follow-up with regulatory bodies later this year.

Moderna’s bivalent COVID-19 vaccine booster, mRNA-1273.214, generated nearly twice the protective antibodies against Omicron.
Adults living with HIV were more likely to have a breakthrough COVID-19 infection after vaccination, suggesting a need for additional vaccine doses in this population.

A new study from the CDC underscores the importance of staying up to date on COVID-19 vaccines and boosters, showing a rapid drop in vaccine effectiveness among children during the Omicron wave that increased with booster doses.
Pfizer-BioNTech Reports Strong Immune Response to Booster Dose in Children 6 Months to Under 5 Years
Companies say the 3 doses of their vaccine meet all immunobridging criteria required for the Emergency Use Authorization (EUA).

The CDC’s COVID-19 booster dose recommendation for children 5 and older comes a few days after the FDA’s authorization

Today, the International AIDS Vaccine Initiative (IAVI) and Moderna announced they will soon launch a phase 1 clinical trial for their HIV vaccine candidate, administered with mRNA technology to develop broadly neutralizing antibodies.

Public health and school officials need to consider strategies now.
A new study in the United Kingdom provided evidence in support of a fourth dose of COVID-19 vaccines, showing a strong boost in immunity.

A third, “booster,” dose of the Pfizer-BioNTech COVID-19 vaccine is now FDA-approved for children 5-11 years old.

The company is utilizing this platform to address new COVID-19 variants, combination vaccines, and potentially prevent HIV.
The company's investigational vaccine, which is in a phase 3 study, is an example of its commitment to addressing these viruses.

At 1 year after COVID-19 infection, children’s neutralizing antibodies only differed by vaccination status.

Despite a significantly slower than expected COVID-19 vaccine rollout, Novavax reports early 2022 as its first profitable quarter.
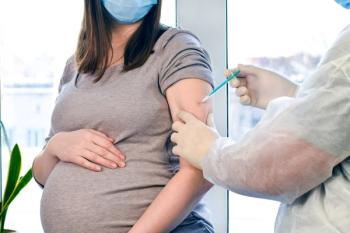
The higher the gestational age when pregnant women received a COVID-19 vaccine, the higher the serological titers at birth.

Active immunization of pregnant women with investigational vaccine protected infants from RSV from birth through at least 6 months.

Children living with HIV had a similar immune response to mRNA COVID-19 vaccination as children without HIV.

A member of the herpes family, Epstein-Barr virus is the primary cause of infectious mononucleosis and may also cause certain cancers and autoimmune diseases.

The federal agency revised the vaccine’s Emergency Use Authorization after an analysis of the risk for thrombosis with thrombocytopenia syndrome (TTS).

Medicago’s plant-based vaccine, Covifenz, was 69.5% effective against symptomatic COVID-19 infection and 78.8% effective against moderate-to-severe disease.