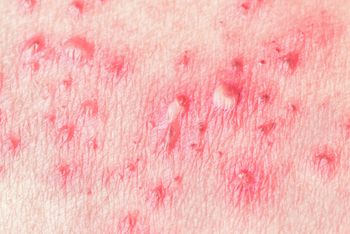
The only way individuals can reduce their risk of getting shingles is to get vaccinated; luckily, a new vaccine has recently been approved by the FDA.

The only way individuals can reduce their risk of getting shingles is to get vaccinated; luckily, a new vaccine has recently been approved by the FDA.

William Schaffner, MD, discusses efforts to develop a universal flu vaccine.

In case you missed them, we've compiled the top five infectious disease articles from this past week.

Richard Haupt, MD, MPH, discusses the process for developing a vaccine.

We’ve compiled the best ways to fight back against colds and flu viruses this holiday season.
A new study conducted by University of Texas Health Science Center at Houston investigators finds that male virgins can still acquire HPV, underscoring the need for vaccination.

More states are reporting widespread flu activity as CDC researchers up their annual estimate of flu deaths around the world.

William Schaffner, MD, explains the strengths and weaknesses of the flu vaccine.
Individuals with an egg allergy do not need to avoid the annual flu shot.

In case you missed them, we've compiled the top 5 articles from this past week.

Richard Haupt, MD, MPH, discusses the different pharmaceutical companies’ efforts to develop a C. difficile vaccine.

New CDC study yields higher estimate of seasonal flu-related deaths worldwide.

As new flu deaths are reported throughout the United States, a pair of studies highlights some of the strengths and weaknesses of the flu shot.

William Schaffner, MD, shares stories to motivate patients and health care providers to receive the flu vaccine.
Five outbreaks of hepatitis A continue to beat down on several states; can addressing the homelessness crisis prevent future outbreaks from happening?

Predicting the next infectious disease outbreak may be possible by analyzing trends on Twitter and Google.

Richard Haupt, MD, MPH, explains how Pfizer is addressing the need for a C. difficile vaccine.

Stay up-to-date on the latest infectious disease news by checking out our top 5 articles of the week.
A pair of studies recently conducted as part of the Human Vaccines Project are giving new insight into the human immune system and how researchers can develop better vaccines.
Investigators are hopeful that the global vaccine candidate will prevent a wide range of strains of the virus.

Following a severe flu season in the Southern Hemisphere and mounting research that egg-based flu vaccines don’t offer enough protection, experts are warning that the Northern Hemisphere may be in for a tough flu season.

The results of 3 phase 1 clinical trials have shown that an investigational Zika purified inactivated virus vaccine was well-tolerated and induced an immune response in adult participants.

William Schaffner, MD, addresses common misconceptions regarding the influenza vaccine.

Stay up-to-date on the latest infectious disease news by checking out our top 5 articles of the week.
A new discovery could lead to a more effective live attenuated influenza vaccine offering extra protection to young children and the elderly.